Understanding Types of Testosterone Injections
Since its inception in 1930, Testosterone Replacement Therapy (TRT) has been a preferable choice for most men and even transgender males to treat their hypogonadism (low testosterone level). TRT, or Hormonal Replacement Therapy, as it is sometimes called, is treated with prescription injections, oral tablets, pellets, patches, and gels to increase the testosterone level reduced by hypogonadism. While the use of TRT injections in treating this condition has proven to be the most common and effective over time, many doctors and patients have continued to find better ways of administering the treatment. This is why the two testosterone injection modes used in Testosterone Replacement Therapy (Subcutaneous and Intramuscular) have often been juxtaposed to measure the effectiveness of one over the other and to guarantee better health conditions for those taking the treatment.
This blog post will attempt a compThis blog post will attempt a comparative analysis of the Subcutaneous and Intramuscular injections and how they are both administered to improve the testosterone levels of hypogonadism patients. Patients’ hematocrit response to the Testosterone Replacement Therapy will equally be considered. However, before any of these, an exposition of the two injections used in TRT will be studied.
INTRAMUSCULAR AND SUBCUTANEOUS INJECTIONS
When men with low testosterone levels are symptomatic from primary or secondary hypogonadism, they are mostly administered testosterone injections. Of the two injections normally administered, the Intramuscular (IM) testosterone is more popular and also the more excruciating of the two. This is because testosterone administered via Intramuscular (IM) injections, as the name goes, have to be injected into the muscle of the patient. For most patients, the Intramuscular injection areas are usually somewhere around the hips, the buttocks, the thigh muscles, and the upper arm. This is to ensure that it goes deep into the muscles of the patient.
On the other hand, the second testosterone injection modal has begun to gain some popularity as more and more patients are seeking a less painful way to get their Testosterone Replacement Therapy. The Subcutaneous (SubQ) testosterone injection is much preferred because it gets the job done without having to go deep into the muscles of the patient. This testosterone injection is inserted into the fat just a little beneath the skin of the patient. The administering areas for this injection include the abdominal areas, the arms, and the front of the thigh. This modal doesn’t leave the patient with an excruciating feeling after the injection.
COMPARATIVE STUDY OF SUBCUTANEOUS AND INTRAMUSCULAR METHODS
Apart from the discomforting weekly or biweekly experience of patients administered the Intramuscular (IM) testosterone injection and the seemingly painless experience of those administered the Subcutaneous (SubQ) testosterone, there seem to be other qualities and distinctions between the two injection modals. Their similarities and distinctions are what most patients and providers have used in evaluating which is more effective when it comes to doing the work they were administered to do.
While Intramuscular (IM) testosterone injections are effective, they pose some difficulty when it comes to their self-administration by patients. This is why most patients are often advised not to self-administer to avoid damaging the sciatic nerve. However, with SubQ, patients can easily self-administer testosterone without any fear of damaging a potential nerve in the body.
As regards the efficacy of both the SubQ and the IM testosterone injections, some men and even transgender males have noted slight changes in the use of one over the other. A 2019 study carried out on transgender males after the first six months of the TRT SubQ, and IM testosterone injections by Oregon Health and Science University stated that the efficacy of the Subcutaneous (SubQ) testosterone is about the same as that of the Intramuscular (IM) testosterone. The study carried out by this university also accounted for lesser adverse effects of the SubQ than the IM. This result, together with a similar result from a study carried out on transgender males by Transgender Health, accounted for the equal efficacy of the SubQ and IM testosterone injections.
The study by the Oregon Health and Science University comparing Subcutaneous Testosterone to Intramuscular Testosterone in gender affirming care of transgender Male Adolescents also provided for the Quality of Life (QOL) of adolescent transgender males who were administered either of the two injection modals. According to this study, the males who were administered the SubQ reported a higher and better quality of life and more satisfactory life and testosterone levels than those administered the IM testosterone injection because the SubQ is painless and easier to self-administer than the IM.
The minimal discomfort and less serum trough fluctuations of the SubQ is another major distinction between the IM and the SubQ approach. Most patients who get the IM injections administered into their muscles experience more testosterone peaks than those who are administered the SubQ. The typical testosterone level for a healthy adult male is between 300 and 1000 nanograms per deciliter (ng/dl), so with the SubQ injections, the patient’s testosterone level rises but remains stable and within the healthy range.
One other distinction between the SubQ and the IM testosterone injections is that the SubQ is less expensive for patients than the IM. This is because the Subcutaneous (SubQ) testosterone injection requires lesser clinic visits than the IM. When the hospital, clinic, or service provider visits are few, the healthcare costs are subsequently and significantly reduced. Most SubQ patients self-administer the testosterone in the comfort of their homes, and they don’t have to visit the clinic weekly like most Intramuscular (IM) testosterone patients do.
Hence, due to the above similarities and differences between the IM and SubQ approaches to TRT, more and more people have begun to opt for SubQ injections than the IM injections, which cause huge discomfort.
HOW TO ADMINISTER SUBCUTANEOUS VS. INTRAMUSCULAR INJECTIONS
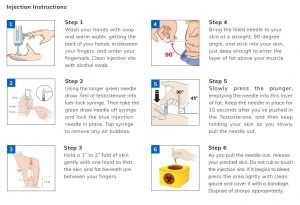
To safely administer either the Testosterone cypionate or the Testosterone enanthate subcutaneously or intramuscularly, you have to prepare a clean and comfortable area. What you would need to set up is your vial of testosterone (cypionate or enanthate), an 18 gauge filter needle, a 25 gauge injection needle with a fixed syringe, some alcohol wipes, and a bin for disposing of needles. Also, ensure you wash your hands before/after administering the injection.
For the subcutaneous injections, do the following:
- Prepare the body area that is to be injected (the abdominal area, fronts of the thigh, arms, and buttocks).
- Snap a twig to break the ampoule at the neck of the enanthate (if that’s what you would be using). If you are using cypionate, remove the vial and open it.
- Examine your syringe and needles to ensure that they have not been pre-opened or damaged before you open them up.
- Use the 18 gauge injection needle to draw your testosterone by screwing the needle to the end of the syringe. Then insert the needle into the testosterone and pull the plunger till you get the prescribed dose.
- Replace the 18 gauge needle with the 25 gauge needle but do not remove the cover.
- Clean the injection site with the alcohol wipe and let it dry.
- Then at a 45-degree angle, inject the site (not so deep, just beneath the skin).
- Once done, wipe with an alcohol swab.
For Intramuscular injections, note that the injection areas are the muscles around your hips and your buttocks. So, once you are done drawing the testosterone, wipe the sin with an alcohol slab and do the following;
- Stretch the area with your hands.
- Inject the site at a 90-degree angle.
- Wipe the surface with the alcohol swab.
HEMATOCRIT RESPONSE FOR PATIENTS ON TRT
While Testosterone Replacement Therapy is good for increasing testosterone levels, it also has one major side effect. TRT patients are prone to have increased hematocrit and hemoglobin, which, when not adequately checked, could lead to cardiovascular deaths and thrombosis. The abnormal increase of the red blood cells (hematocrit) is called Erythrocytosis. Hence, knowing the healthy and normal hematocrit levels of adult males (between 40 and 54%) and females (between 36 and 48%) is helpful in keeping things in check. Also, to be on the safer side, TRT patients are advised to regularly check their hematocrit levels.
Phlebotomy, or the removal of some of your blood to remove excess red blood cells and improve blood flow, may be used to treat primary erythrocytosis. The underlying cause of secondary erythrocytosis is treated. This treatment may comprise the following:
- Oxygen for persons with low oxygen levels
- Advice to stop smoking
- Having a surgical procedure to to remove tumors
- Reduction or stoppage of medicines that are causing the erythrocytosis
- Treatment of any disorder linked to the low oxygen levels
How To Administer Subcutaneous Injections
FAQ
CONCLUSION
Testosterone injections are commonly used to treat sexual dysfunction in men and postmenopausal symptoms in women who have low testosterone levels. Testosterone injections are also used by transgender men and nonbinary people as part of masculinizing therapy. Many people find testosterone injections to be safe, although they do have negative effects. Overall, both the Subcutaneous (SubQ) and the Intramuscular (IM) testosterone injections are used by many patients in the world. The preference for one over the other does not change the fact that both are of the same efficacy. This is why the SubQ is only seen as a considerable alternative to the Intramuscular injections and not a replacement for the IM.
Although most studies have stated that lesser dosages in SubQ have fewer adverse effects than the higher dosages in IM, the two injections should be administered with equal caution and with the prescribed dosage.
If you’re experiencing symptoms of low testosterone and want to learn more about treatment options, contact HRT Doctors Group today! Our team specializes in hormonal therapies and can help you find the right solution for your needs. We will do our best to help you find your way to hormone balance and well-being.